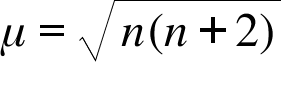The highest value of the calculated spin-only magnetic moment (in BM) among all the transition - YouTube

Q4)Calculate spin only magnetic moment of the following ions in aqueous state: (a) Mn2+ (b)Cr3+ (c)Co3+ - Chemistry - Aldehydes Ketones and Carboxylic Acids - 16918049 | Meritnation.com

58. If spin only magnetic moment of vcl, is 1.73 BM then correct formula is :- * vci, (2) VCI, X (3) VCI, (4) VCI, 53 -1.732. 3ds socarz?

Write the name of two metal which used in maximum composition in mischmetal. Calculate the value of the magnetic moment of V^{+2}

Write the spin only formula and give the unit of magnetic moment - Chemistry - Structure of Atom - 15663443 | Meritnation.com
![The spin only magnetic moment value of `[MnBr_(4)]^(2-)` ion is `5.9BM` On the basis of `VBT` - YouTube The spin only magnetic moment value of `[MnBr_(4)]^(2-)` ion is `5.9BM` On the basis of `VBT` - YouTube](https://i.ytimg.com/vi/no_ZGBD689M/maxresdefault.jpg)
The spin only magnetic moment value of `[MnBr_(4)]^(2-)` ion is `5.9BM` On the basis of `VBT` - YouTube
![The complex with spin only magnetic moment of ~4.9 BM is:a)[Fe(H2O)6]2+b)[Fe(CN)6]3-c)[Fe(CN)6]4-d)[Fe(H2O)6]3+Correct answer is option 'A'. Can you explain this answer? - EduRev Chemistry Question The complex with spin only magnetic moment of ~4.9 BM is:a)[Fe(H2O)6]2+b)[Fe(CN)6]3-c)[Fe(CN)6]4-d)[Fe(H2O)6]3+Correct answer is option 'A'. Can you explain this answer? - EduRev Chemistry Question](https://edurev.gumlet.io/ApplicationImages/Temp/9569037_ab072f64-2e6d-4920-b550-ecfc86b0a8de_lg.png)
The complex with spin only magnetic moment of ~4.9 BM is:a)[Fe(H2O)6]2+b)[Fe(CN)6]3-c)[Fe(CN)6]4-d)[Fe(H2O)6]3+Correct answer is option 'A'. Can you explain this answer? - EduRev Chemistry Question



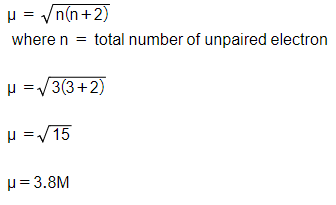


![ANSWERED] 140 The calculated spin only magnetic mom... - Physical Chemistry - Kunduz ANSWERED] 140 The calculated spin only magnetic mom... - Physical Chemistry - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20201001105030189875-2167239.jpg)